 Based on the in vivo concentrations, we investigated the effect of nicotine on NAD + regulation in vitro by using an appropriate range of concentrations of 10–20 ng/mL of nicotine in cultured cells. The nicotine content in the liver and brain were about 15 ng/g, and about 0.25 ng/g in the serum (Supplementary Fig. We directly measured the nicotine content of the aged tissue using LC-MS. To further validate the binding and biological function of SIRT1 to NAMPT via nicotine, we calculated the exact concentration of nicotine in the tissues during this chronic administration. It has been discovered that minuscule amounts of nicotine, as an activator of NAD + biogenesis, can markedly improve NAMPT activity and NAD + synthesis, leading to improved glucose metabolism and cognitive function, as well as aging symptoms, in male mice. Consequently, we hypothesized that nicotine may play a role in the NAD + salvage pathway. Furthermore, the NAD + salvage pathway is conserved across a variety of species, including bacteria, plants, yeasts, and mammals 20. Interestingly, nicotine is a secondary metabolite of the NAD + biosynthesis and the NAD + pathway is also coordinately regulated with nicotine biosynthesis 20, 21. Therefore, the enhancement of NAMPT activity could be a potential strategy for preventing aging and age-associated diseases arising from NAD + decline 11, 18, 19. Moreover, supplementing the high active extracellular NAMPT (eNAMPT) also increases NAD + biogenesis and aging-related symptoms 15, 16, 17. Consequently, the NAMPT enhancers can effectively increase NAD + levels and prevent neuronal degeneration 12, 13, 14. As NAMPT activity decreases with age, it results in NAD + depletion and cognitive impairment 11. In mammalian cells, most NAD + precursors were “reclaimed” to NAD + via the rate-limiting enzyme nicotinamide phosphoribosyl transferase (NAMPT) of the salvage pathway. Previous studies showed that supplementing NAD + precursors, such as nicotinamide mononucleotide (β-NMN), nicotinamide riboside (NR), nicotinamide (NAM), or Niacin (NA), were efficient for anti-aging 8, 9, 10. To counteract this, two strategies have been proposed: reducing the activity of NAD +-consuming enzymes 6, 7 and increasing the biosynthesis of NAD + by supplying NAD + precursors and activating NAD + synthetic enzymes. However, with advancing age, the equilibrium of NAD + homeostasis is impaired, leading to a considerable decline, correlating with age-related defects 1, 2, 3, 4, 5. Nicotinamide adenine dinucleotide (NAD) homeostasis is organized and coordinated in many physiological statuses. Collectively, these findings provide evidence for a mechanism by which low-dose nicotine can activate NAD + salvage pathways and improve age-related symptoms. Additionally, nicotine ameliorated cellular energy metabolism disorders and deferred age-related deterioration and cognitive decline by stimulating neurogenesis, inhibiting neuroinflammation, and protecting organs from oxidative stress and telomere shortening. 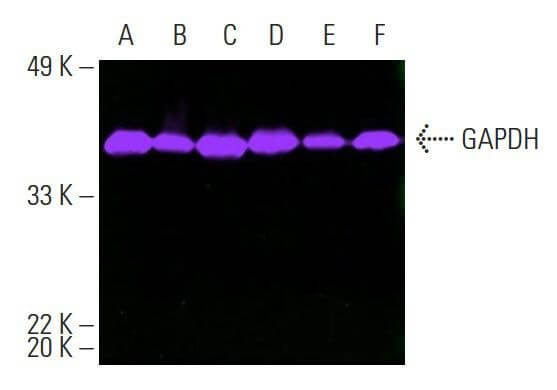 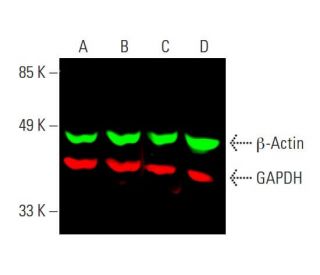
18F-FDG PET imaging revealed that nicotine is also capable of efficiently inhibiting glucose hypermetabolism in aging male mice. Here we find that, independent of nicotinic acetylcholine receptors, low-dose nicotine can restore the age-related decline of NAMPT activity through SIRT1 binding and subsequent deacetylation of NAMPT, thus increasing NAD + synthesis. Nicotine, a metabolite of the NAD + metabolic pathway, has been found to possess anti-inflammatory and neuroprotective properties, yet the underlying molecular mechanisms remained unknown. Imbalances in NAD + homeostasis have been linked to aging and various diseases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
